Using (2S, 4R) - Boc-trans-4-hydroxy-L-proline methyl ester (8) as the starting material, through oxidation, reduction and deoxyfluorination, the stereocenter of its secondary alcohol undergoes double inversion to produce trans-4-fluoro-L-proline (13); Subsequently, the stereoselectivity was determined by the Seebach method α- The carbonyl position is introduced into a three-carbon compound to synthesize 15; Finally, deprotection and cyclization form the bicyclic core of bicyclopyrrolitol, and the final product 1 is obtained after reduction.

Org. Process Res. Dev. 2022, 26, 2839?2846
1、 Synthesis of trans-4-fluoro-L-proline (13)
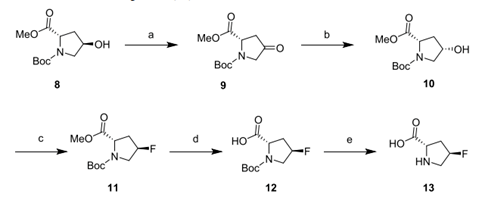
A. Compound 9 is obtained from 8 by oxidation;
B. Keto reductase (KRED) PK076 is used to convert and reduce the ketone group in 9 to obtain 10. The conversion rate can reach 99.5%, the enantiomeric ratio is 98.7:0.8, and the enzyme load is 10%. In addition, the efficiency of biotransformation reached more than 99% in the 100g level experiment, and the enantiomeric ratio reached 99.3:0.7 (98.6% de). Pure product 10 was obtained by crystallization with n-heptane/MTBE system, and the yield was 87%.
c&d. Use trifluoromethanesulfonic anhydride and Et3N ・ 3HF to carry out two-step deoxyfluorination of 10. The elimination reaction will occur in the fluorination reaction to produce olefin by-products, which can be removed by KMnO4 oxidation. Other residual impurities are also removed in the downstream process. The separation purity is>99%, the mass fraction of the product after crystallization is>99%, and the yield is 71%.
2、 Synthesis of compound 1 from 13
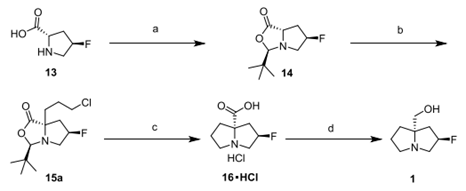
A. Use 1.2 eq formaldehyde and 4.5 eq trimethyl orthoformate as reaction reagents to generate 14 at 45 ℃. The yield was 91%.
B. Stereoselectivity with electrophilic haloalkanes α- Alkylation reaction. As shown in the following table, a series of optimization screening has been carried out for different haloalkanes and catalytic conditions:
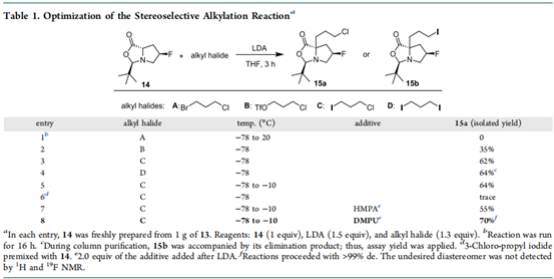
Finally, it was found that the yield of iodochloropropane was 70% under the catalysis of DMPU (2 eq.), which was the optimal condition.
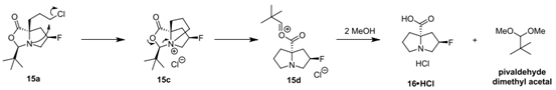
C. Reflux 15a in methanol for 16 hours to produce 16 ・ HCl. It is easy to separate the required products through acetone crystallization. In terms of mechanism, it is assumed that the reaction is initiated by intramolecular alkylation to form quaternary ammonium salt 15c. After acetal decomposition with methanol, the acetal ester breaks into the required products 16 ・ HCl and neopentaldehyde dimethyl acetal 15d.
D. Reflux 16 ・ HCl and (2 eq.) BF3 ・ OEt2 and (3 eq.) NaBH4 in THF to produce the final product 1, which is light yellow oil with a separation yield of 84%.
The process has been successfully applied to the synthesis of compound 1 in kilogram scale with a total yield of 40% and selectivity of (99% ee and de).







